All published articles of this journal are available on ScienceDirect.
Health Significance of the COVID-19 Pandemic in People with Primary Ciliary Dyskinesia, A National Cohort Study
Abstract
Introduction
During the COVID-19 pandemic, Danish health authorities implemented nationwide lockdowns and recommended strict preventive measures, particularly for individuals with chronic lung disease. This national study evaluated the health impact of COVID-19 in people with Primary Ciliary Dyskinesia (pwPCD) compared with the Danish background population.
Methods
Genetically confirmed pwPCD of all ages who consented were included. Data from March 2019 to March 2022 covered SARS-CoV-2 test results, vaccination status, hospitalization, COVID-19 related morbidity, z-scores of forced expired volume in one second; z-FEV1, and mortality. SARS-CoV-2-positive participants completed a retrospective symptom-score questionnaire. Descriptive statistics and linear mixed-effects models were applied.
Results
Of 124 Danish pwPCD, 79 (64%) participated (44% male, median age 24.3, range 0.4-71 years). SARS-CoV-2 positivity was significantly lower in pwPCD (20%) compared to the background population (58%) (p<0.0001). Most infections (94%) were mild. Hospitalization and mortality rates were comparable (0.8% versus 0,8%; 0% versus 0.2%, respectively). Vaccination acceptance was high (92% versus 82%), though it varied by age. z-FEV1 remained stable pre-pandemic (0.02/year, p=0.65) and during the pandemic (-0.03/year, p=0.44), with only a clinical negligible difference (-0.09/year, p=0.024).
Discussion
COVID-19 had minimal health impact on pwPCD, with fewer infections, high vaccination adherence, and stable lung function. These findings contrast with concerns about severe outcomes in chronic lung disease and may reflect adherence to preventive measures.
Conclusion
Stringent health measures and proactive care may effectively protect pwPCD during pandemics. Similar strategies could be applied in future pandemics to protect pwPCD or other chronic lung diseases.
1. INTRODUCTION
Chronic respiratory diseases are well-established risk factors for severe COVID-19 outcomes associated with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).[1–3]. However, data on specific conditions and their impacts remain limited. In most cases, SARS-CoV-2 infection results in mild to moderate illness, with recovery typically occurring without the need for specialized medical intervention such as emergency care, hospitalization, or intensive care. Contrarily, research indicates that individuals with chronic respiratory conditions - including cystic fibrosis and chronic obstructive pulmonary disease - are at increased risk of severe illness, admission to intensive care units, and higher mortality rates[4–7].
Primary ciliary dyskinesia (PCD) is a rare genetic disorder with an estimated minimum prevalence of 1 in 7,554[8]. It is characterized by defective motile cilia function, leading to chronic upper and lower airway disease, impaired lung function, and structural lung damage[9]. The severity of PCD varies considerably among individuals, ranging from mild respiratory symptoms to severe, irreversible lung disease with reduced lung function and bronchiectasis[10–13]. The clinical presentation and outcomes of COVID-19 in individuals with PCD remain poorly characterized and may differ due to the heterogeneous nature of the disease[14].
The entire Danish cohort of people with PCD (pwPCD) is monitored centrally at the National Danish PCD Center in Copenhagen, providing a unique opportunity to assess the health impact of the COVID-19 pandemic in this population.
The objective of this study was to evaluate whether the strategy implemented at our PCD center effectively mitigated the risk of severe COVID-19-related pulmonary disease among these patients.
We hypothesized that pwPCD demonstrated high adherence to our guidelines, likely driven by both patient and provider concerns regarding severe COVID-19 lung disease, and that our stringent safety measures conferred a protective benefit during the pandemic. This study aimed to report SARS-CoV-2 infection rates, morbidity, mortality, and longitudinal lung function stability in Danish individuals with PCD during the national lockdown period.
2. METHODS AND ANALYSES
2.1. Design and Study Population
This was a single-center, national observational cohort study conducted at the National Danish PCD Center, Copenhagen University Hospital. The study included all individuals of any age with genetically confirmed PCD who provided written informed consent.
Exclusion criteria were the absence of signed informed consent or the lack of genetic confirmation of the PCD diagnosis.
Data were extracted from participants’ electronic medical records, and COVID-19 symptom data were retrospectively collected through a symptom-score questionnaire sent to patients who had at least one positive SARS-CoV-2 test.
2.2. Study Procedures and Questionnaire
At the onset of the pandemic in Denmark, national authorities implemented strict lockdown measures beginning on 12 March 2020, including social restrictions and stringent hygiene recommendations. These restrictions were intermittently lifted and reintroduced with varying degrees of stringency until March 2022. Due to their presumed vulnerability, Danish pwPCD were advised to adhere to strict isolation measures similar to those recommended for individuals with other chronic respiratory diseases during the entire pandemic[15–17]. Significant resources were allocated at the National Danish PCD Center to implement safety protocols aimed at protecting both healthcare workers and patients, as pwPCD were classified as “high-risk patients”. Aerosol-generating procedures, such as lung function test and laryngeal suction/expectoration, were performed daily at the center. Safety measures, therefore, included mandatory negative polymerase chain reaction (PCR) testing for the patient or their parent (for children under 5 years), conducted within 48 hours before a visit. Protective equipment, including face masks, was required for healthcare workers, parents, and patients during examinations. For aerosol-generating procedures, healthcare workers additionally wore long-sleeved protective gowns. To compare data between the pandemic period and the preceding year, we collected demographic data and comprehensive healthcare data from March 2019 to March 2022, including SARS-CoV-2 testing, vaccination status, unplanned hospital visits or admissions, COVID-19-related morbidity, and lung function measurements (z-scores of forced expired volume in one second; z-FEV1).
Participants who tested positive for SARS-CoV-2 during the study period were asked to complete a self-reported symptom-score questionnaire. This questionnaire included items on symptoms, illness duration, healthcare visits (emergency department, general practitioner), and hospitalizations. For children under 15 years, the questionnaire was completed by the parents or guardians, whereas individuals aged 15 years and older completed the questionnaire themselves. Data regarding emergency visits or hospitalizations were verified in patients’ medical records.
2.3. Outcomes
The primary outcomes of the study included: the number of SARS-CoV-2 tests performed during the study period; the ratio of positive SARS-CoV-2 tests per patient; SARS-CoV-2 vaccination status; COVID-19 related hospitalization (categorized as: emergency room only, hospital admission; intensive care unit admission; and number of days hospitalized); morbidity data (symptoms during COVID-19, duration of symptoms, and need of general practitioner visits); lung function assessed as z-FEV1, calculated using the global lung initiative (GLI) 2012 dataset [18, 19]; and mortality rates. As FEV1 was expressed as a z-score, adjustments were made for sex, age, and height. For vaccination status and SARS-CoV-2 positive patients, differences were analysed according to age.
Although specific SARS-CoV-2 variants were not available, dates of positive PCR tests were correlated with publicly available data on predominant variants in Denmark from Statens Serum Institut, the national disease surveillance authority [15].
2.4. Ethics
The study was approved by the Center for Research and Innovation in the Capital Region of Denmark (P-2021-541), including permission to contact families outside the clinic (R-21041716). As a non-interventional observational study, additional approval from the ethics committee was not required. The study adhered to the principles of the Helsinki Declaration.
Patients were recruited either by telephone or during routine outpatient visits between July 2021 and June 2022. Participants aged 15 years and older provided informed consent before inclusion, while parents or legal guardians provided consent for children under 15 years of age. Parents or legal guardians of adolescents aged 15-17.9 years were informed about the study.
2.5. Background Population
Data on SARS-CoV-2 testing, COVID-19-related hospitalizations, vaccination status, and mortality rates in the general population were obtained from publicly available sources provided by Statens Serum Institut [15]. This dataset served as a benchmark for evaluating outcomes in pwPCD.
2.6. Statistical Analyses
The size of our study cohort was opportunistic, as all eligible pwPCD who consented to the study were included. Therefore, no formal power calculation was performed. Missing data were assumed to be missing at random, including non-consent, incomplete lung function data, and questionnaires.
Statistical analyses were primarily descriptive, including counts, ratios, percentages, and t-tests for group comparisons. To describe the SARS-CoV-2 infection rates among pwPCD, we calculated the proportion of patients who tested positive during the study period relative to the total number of included patients.
To assess changes in lung function (z-FEV1) before and during the lockdown period, we employed a linear mixed-effects model, adjusting for measurement timing. Patient-ID and age (slope) were included as random effects, allowing each patient to have a unique intercept and slope of lung function over time. This approach enabled evaluation of longitudinal lung function trajectories and determination of any significant lockdown-related effects. The mixed-effect model accounted for varying numbers of lung function measurements across patients. Analyses were performed for the entire cohort and stratified by age groups: children (< 18 years) and adults (≥18 years), as COVID-19 appeared to affect children less severely than adults.
An unstructured covariance structure was applied to allow for each pair of observations to have its own correlation and separate variances for intercept and slope.
All statistical analyses and data visualization were conducted using R Studio (version 4.2.2), SAS Enterprise Guide (version 7.1), and Microsoft Excel (2016).
3. RESULTS
The study included 79 individuals aged 0.4 to 71.0 years, representing 64% of the 124 genetically verified PCD patients monitored at the Danish PCD Center in Copenhagen. Fig. (1) illustrates a flow diagram of the inclusion of patients and grouping during the study. At the beginning of the pandemic, the median age of the cohort was 24.3 years (interquartile range [IQR ]17.3; 34.0), with only five patients (6%) aged above 50 years. Forty-four participants (56%) were females. Demographic characteristics are presented in Table 1.
| Patients included in the study, N (% of total Danish cohort) |
79 (64%) |
|---|---|
| Age (years) | 24.3 (0.4; 71.0) |
| Children (0-17.9 years) | 22 (28%) |
| Adults (above 18.0 years) | 57 (72%) |
| 50 years and above | 9 (11%) |
| Sex | |
| Male | 35 (44%) |
| Female | 44 (56%) |
| BMI, all (bodyweight/[height in meter]2) | 22.3 (15.3; 46.6) |
| BMI, 16 patients with COVID (bodyweight/[height in meters]2) | 19.5 (15.7; 27.0) |
| Genetic variants associated with PCD | |
| • CCDC39+CCDC40 (often severe) | 17 (21.5%) |
| • DNAH11+HYDIN (often mild) | 13 (16.5%) |
| • DNAI1+DNAH5 | 20 (25.3%) |
| • Others | 29 (36.7%) |
| Bronchiectasis verified by HRCT, all patients | 69 (87%) |
| Bronchiectasis verified by HRCT, 16 patients with COVID | 13 (81%) |
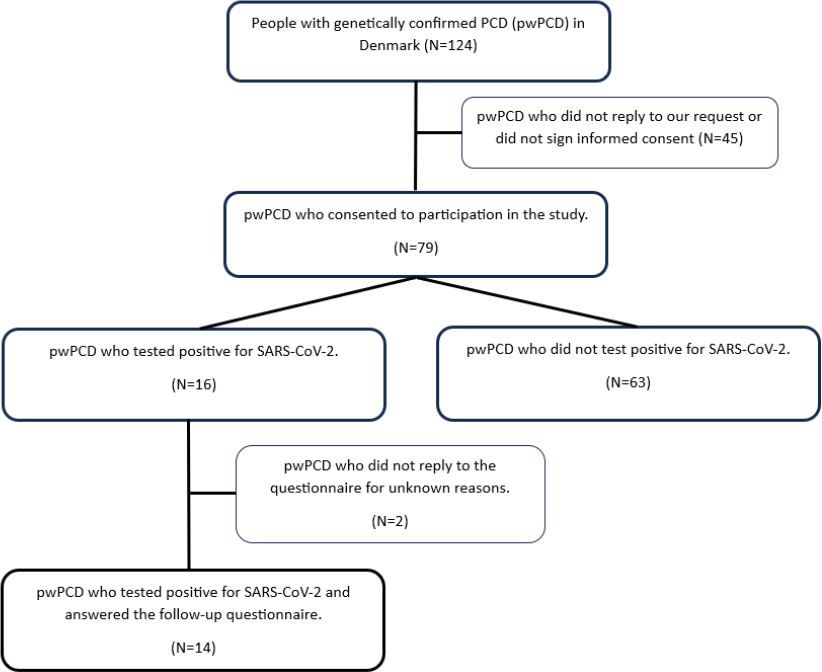
Flow diagram showing the selection of patients in the study and grouping within the cohort.
3.1. Incidence and Severity of COVID-19
During the study period, each pwPCD underwent between one and 76 officially registered SARS-CoV-2 PCR tests. In total, 1718 PRC tests were conducted, with a median of 19 tests per patient (IQR: 12-27). Of all tests, 16 (0.9%) were positive, and no patients had more than one positive test. Overall, 16 out of 79 pwPCD (20%) tested positive for SARS-CoV-2. This was significantly lower than the 58% infection rate observed in the general population, corresponding to a difference of 38% (95%CI: 27.6% to 44.2%, P < 0.0001).
No pwPCD tested positive for the index variant of SARS-CoV-2 during the initial phase of the pandemic when complete lockdown measures were in place. Additionally, none of the patients tested positive in the subsequent months, during which both children and adults with chronic lung diseases were advised to adhere strictly to the safety guidelines, despite the gradual normalization of social activities in the country. The first positive test occurred in December 2020, when the Alpha (B.1.1.7) variant was the predominant strain in Denmark. Five patients tested positive between December 2020 and July 2021, where the dominant variants were Alpha and Delta (B.1.617.2, AY). The remaining 11 patients all tested positive between November 2021 and March 2022 during the time dominated by the very contagious, but in general milder disease-causing variant, Omicron (B.1.1.529, BA).
Hospitalization rates among pwPCD infected with SARS-CoV-2 were low: only one patient required a two-day hospital stay, and none required intensive care. This corresponds to 6.3% of the infected patients in the study cohort, 1.2% of the entire study cohort. To our knowledge, no pwPCD from the non-responding group were hospitalized due to COVID-19, i.e., only one of the 124 Danish pwPCD that were hospitalized (0.8%). This patient was a female in her mid-twenties, vaccinated against SARS-CoV-2, likely infected with the Omicron variant. Her genetic variant was DNAH11, considered mostly a mild variant; her pre-infection lung function was normal (z-FEV1: +0.56); like most pwPCD, she had bronchiectasis confirmed by high-resolution computed tomography (HRCT), and a BMI of 27.
In comparison, also 0,8% of the background population required hospitalization due to COVID-19, and 2% of the background population was admitted to the hospital with a positive COVID-19 PCR test. Approximately 60-70% of those admitted due to COVID-19 were aged ≥ 65 years. The remaining had at least one comorbidity such as obesity, chronic lung disease, diabetes, cardiovascular disease, or immunodeficiency. Moreover, while the mortality rate for COVID-19 in the general population was 0.2%, no deaths occurred among the pwPCD (P = 0.68).
Among the 16 pwPCD who tested positive for SARS-CoV-2, 14 (88%) completed the follow-up questionnaire. Of these, 1 patient (7%) reported no symptoms, while the remaining 86% reported mild symptoms. The median (range) recovery time was 4 (0; 14) days. Only one patient experienced persistent gastrointestinal symptoms after recovery. Rates of symptoms are shown in Table 2 and Fig. (2).
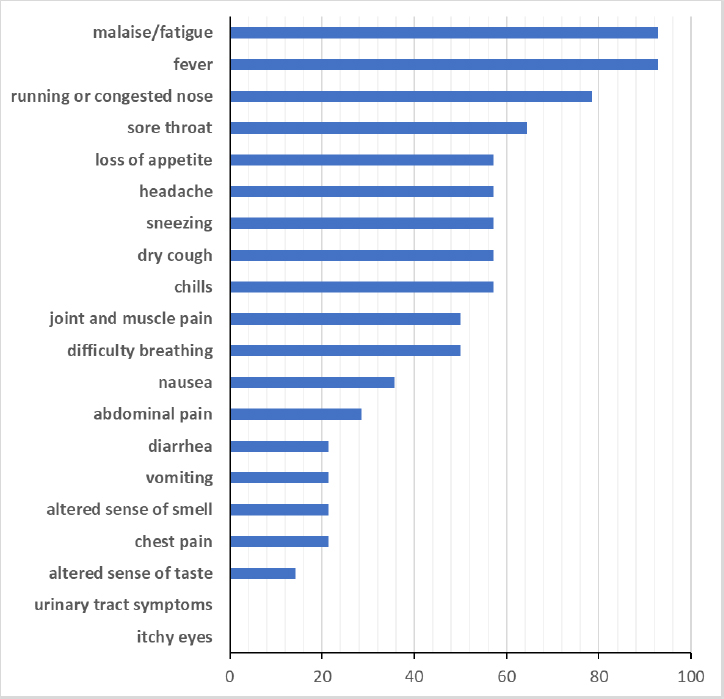
Symptoms reported by 14 people with PCD during infection with SARS-CoV-2
| Answered questionnaire | 14 (87.5%) |
| Sex (female) | 9 (64%) |
| Duration of symptoms (days) | 4 (0; 14) |
| Asymptomatic | 1 (7%) |
| General practitioner visits | 5 (35%) |
| Hospitalization | 1 (7%) |
| Persistent symptoms after recovery | 1 (7%) |
The median (IQR) age at time of positive testing was 18.6 years (13.4; 23.3), with only two preschool children and one patient above age 30 years (53.5 years). Three of the 16 SARS-CoV-2 positive patients had no bronchiectasis; all were children, two with the genetic variant DNAH5 and one with CCDC39.
3.2. Vaccination
A total of 69 out of 79 pwPCD (87%) received the vaccination against COVID-19, a rate higher than the 77% vaccination coverage observed in the background population. Four of the non-vaccinated pwPCD were children under five years of age, who were not included in the vaccination program before November 2022. Only six pwPCD (8%) declined the vaccination offer, meaning that 92% of pwPCD who were offered the vaccination accepted it, compared to 82.3% of the background population above age 5 years. Among children aged 5-11 years (N=4), three children (75%) received the vaccine, whereas only 37% of this age group was vaccinated in the general population. However, in our cohort, three children (27%) aged 12-19 years were not vaccinated, compared to 17% in the general population.
Of the 16 pwPCD infected with SARS-CoV-2, four (25%) were never vaccinated (aged 2, 3, 14, and 30 years), and three (19%) contracted COVID-19 before receiving vaccination. The remaining nine patients (56%) were infected with the Omicron variant despite previous vaccinations.
The median (range) age of pwPCD at the time of their last vaccination dose was 26.3 (9.7; 72.5) years.
3.3. Lung Function and COVID-19
Over the three years from March 2019 to March 2022, we analysed 605 spirometry measurements from 68 pwPCD, representing 86% of the study cohort. Data were missing for 11 pwPCD, five of whom were six years or younger. The median (IQR) number of lung function tests per participant was nine (7; 11); three (2; 4) in the year before the COVID-19 lockdown and two (2; 3) and three (2; 4) in the first and second year of the pandemic, respectively.
Using a mixed effect regression model, the mean (95% CI) z-FEV1 for the group during the year before lockdown was -2.10 (-2.48; -1.73). The lung function remained stable both during the year before lockdown (12 March 2019 to 11 March 2020) and throughout the two subsequent years of the pandemic (12 March 2020 to 11 March 2022). The mean (95%CI) change in z-FEV1 pre-pandemic was 0.02 (-0.20; 0.16) vs. an annual change in z-FEV1 during the pandemic of -0.03 (-0.09; 0.04). This was a nonsignificant difference in progression rates. However, there was a statistically significant, but clinically negligible difference in the mean z-FEV1 of -0.09 (-0.17; -0.01), p = 0.0237 between the pre-pandemic and the pandemic periods. Furthermore, there was no significant difference in mean z-FEV1 or its rate of change between children and adults in the study.
4. DISCUSSION
This study is unique in its inclusion of two-thirds of the entire national cohort of genetically verified pwPCD. It not only evaluates longitudinal lung function data before and during the COVID-19 pandemic. It also integrates critical information on COVID-19 testing, vaccination, and symptoms during SARS-CoV-2 PCR positivity, providing a comprehensive assessment of the pandemic’s impact on this patient population. Our study reveals that the Danish pwPCD, traditionally classified as high-risk patients, exhibited a significantly lower incidence of SARS-CoV-2 infections compared to the general population, with minimal impact on health, as evidenced by stable lung function trends. We found no increased risk of severe COVID-19 outcomes in this group, with hospitalization and mortality rates mirroring those of the background population, however interpreted from a relatively small number of cases. Our results align with a larger questionnaire-based online-study that included self-reported data from individuals with confirmed or suspected PCD[19]. This study also documented primarily mild COVID-19 disease. However, our study differs by including only genetically confirmed cases and by using data extracted from medical records. The observation of stable lung function throughout the pandemic represents a novel finding.
Notably, 94% of pwPCD above age five years were vaccinated, a rate higher than that of the background population (82%). Furthermore, only five patients (6%) were infected before vaccines became available, and the majority (75%) of the infected pwPCD were school-aged children or young adults. These findings suggest that our cohort may have adhered to more rigorous preventive measures, such as strict isolation and enhanced hand hygiene, which could have contributed to lower infection rates, particularly among older pwPCD. It is also plausible that these patients had fewer social contacts due to health-related anxiety, although this was not examined in our study.
In general, Denmark implemented one of the strictest national lockdowns and offered extensive free testing, which may have contributed to the low infection rates among both pwPCD and the general population. The number of SARS-CoV-2 PCR tests per patient was high during the pandemic (median 19, IQR 12-27 in two years). In addition, there was a widespread use of home tests, which are not accounted for in this analysis. The general recommendation during the pandemic was that a positive antibody or home test should be confirmed by an official PCR test. Nevertheless, some infections may have been missed if patients did not follow this recommendation. All patients, except two infants born during the pandemic, underwent at least three official PCR tests.
Those infected generally experienced only mild COVID-19 disease. Factors such as young age, normal BMI, high vaccination coverage, and the predominance of the Omicron variant in those infected may have contributed to these mild outcomes.
The adherence of pwPCD to isolation and precautionary behaviours aligns with findings from other studies, suggesting that individuals with chronic respiratory conditions tend to follow public health guidelines more diligently when enforced by national authorities[20]. However, the downside of strict isolation may include an increased risk of depression[21]. The higher vaccine acceptance among pwPCD was consistent with prior studies indicating increased vaccine willingness within this population[20].
Despite concerns about severe COVID-19 risk in pwPCD, lung function remained stable before and throughout the lockdown period. The finding of stable lung function in the year preceding lockdown is noteworthy and contrasts with the expected decline observed in other studies involving Danish pwPCD[22]. Similarly, lung function stability during the lockdown suggests that reduced exposure to respiratory pathogens in general may have played a role. Supporting this hypothesis, one study reported fewer exacerbations during lockdown[23]. Likewise, another study observed no significant difference in hospitalization rates due to exacerbations among paediatric patients with cystic fibrosis, interstitial lung disease, and PCD, when comparing pre-pandemic and pandemic periods, despite markedly reduced outpatient visits and pulmonary lung function testing[24]. Likewise, fewer routine visits during lockdown did not seem to negatively affect respiratory health in our study.
5. STRENGTHS AND LIMITATIONS
One of the key strengths of this study is the inclusion of 66% of the total national cohort. Also, the access to comprehensive clinical data obtained directly from the medical records is a strength. These data included severity measures such as genetic diagnosis of PCD, lung function, age, height, weight, treatment, and symptoms during hospitalization. This approach provides a more accurate assessment compared to self-reported data as used in previous studies by Pedersen et al.[14, 19, 20]. Additionally, our cohort consists of a well-characterized group of patients with genetically confirmed PCD diagnoses, unlike studies that include individuals with both self-reported, suspected, and confirmed cases[14, 19, 20]. We closely monitored patients throughout the COVID-19 pandemic and collected extensive data longitudinal lung function data during this period. However, a notable limitation of our study is the relatively small sample size of 79 patients, which, although representing the majority (64%) of the Danish PCD population, may limit the generalizability of the findings. In particular, the small number of pwPCD (16) who tested positive for SARS-CoV-2 constrains the interpretation of infection-related outcomes. The remaining 36% of pwPCD in Denmark did not respond to our invitation to participate in the study, leaving uncertainty regarding potential selection bias related to age, comorbidities, geographical location (urban vs rural), or attitudes towards COVID-19. As non-responders did not consent, no systematic comparison was possible. In general, however, pwPCD in Denmark attend the monitoring program regularly regardless of distance to the hospital in Copenhagen, socio-economic status, or health status. Patients with more severe lung disease were highly compliant with scheduled visits. In this cohort, the mean and median ages of those with fewer than half of the expected visits (the non-compliant group, N=15) were not significantly different from those who attended more visits (compliant group, N=64). However, only four children were in the non-compliant group compared to 13 in the compliant group. Likewise, only one patient was older than 40 years in the non-compliant group compared to 11 in the compliant group. The median (IQR) z-FEV1 in the non-compliant group was -1.62 (-2.81; -0.16) compared to -2.51 (-3.47; -1.40) in the compliant group.
The relatively young median age of our cohort, the high socio-economic support in Denmark through a universal healthcare system, and the high vaccination uptake (94%) are all protective factors against severe COVID-19, which may have influenced the study results.
Symptom data were available in the medical records for only one hospitalized patient. The remaining 15 patients with COVID-19 did not consult the hospital or outpatient clinic during their illness. Although some misclassification bias may arise from the retrospective self-reporting, it seems unlikely that patients with severe symptoms would have failed to seek medical care.
CONCLUSION
At the onset of the COVID-19 pandemic, it was uncertain whether pwPCD were at increased risk for severe COVID-19 outcomes. Our findings indicate that pwPCD in Denmark had a lower incidence of reported SARS-CoV-2 infections compared to the background population, and those who were infected experienced only mild symptoms. This may be attributed to the stringent lockdown measures, self-isolation protocols, and extensive testing implemented by the Danish government and the Danish PCD Center in Copenhagen, particularly in the early stages of the pandemic. Notably, no patient tested positive for the index variant, and only five pwPCD were infected with SARS-CoV-2 Alpha or Delta variants. Most infections occurred post-vaccination and were caused by the milder Omicron variant.
Overall, the COVID-19 pandemic had minimal health impact on our cohort of pwPCD, with no mortality and stable lung function throughout the study period.
Future Scope and Prospects
In the event of a future viral pandemic, the strategies employed during the COVID-19 pandemic demonstrate that it is possible to protect vulnerable patient groups from infection. Similar measures could therefore be considered as effective interventions in future pandemics.
Significance of this Study for Society
Evaluating health strategies implemented during the COVID-19 pandemic is essential for informing future public health responses. In our study, pwPCD were not infected with the more severe Alpha-variant of SARS-CoV-2, and most patients remained uninfected until after vaccination, which appears to have protected them from severe COVID-19-related lung disease. These findings underscore the importance of proactive measures and vaccination strategies for safeguarding vulnerable population groups. However, the generalizability of our study results may be limited by the relatively small size of our country and, consequently, the limited sample size.
People with primary ciliary dyskinesia followed the stringent health measure advisements, including social isolation, extensive hygiene measures, and vaccination, leading to low infection rates and stable lung function during the COVID-19 pandemic.
AUTHORS’ CONTRIBUTIONS
The authors confirm contribution to the paper as follows: F.B., M.S., R.M.S., and K.G.N.: Made substantial contributions to the study concept and design; H.C., S.A., and M.P.: Contributed substantially to data collection; M.H. and R.M.S.: Analysed the data; M.S. and R.M.S.: Finalized data collection, analysis, and drafted the manuscript. All authors critically revised and approved the manuscript.
LIST OF ABBREVIATIONS
| BMI | = Body Mass Index |
| CI | = confidence interval |
| COVID-19 | = Coronavirus Disease 2019 |
| FVC | = Forced Vital Capacity |
| IQR | = interquartile range |
| PCD | = Primary Ciliary Dyskinesia |
| PCR | = polymerase chain reaction |
| pwPCD | = People with Primary Ciliary Dyskinesia |
| SARS-CoV-2 | = Severe Acute Respiratory Syndrome Coronavirus 2 |
| z-FEV1 | = z-scores of Forced Expiratory Volume in the first second |
ETHICAL APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the Center for Research and Innovation in the Capital Region of Denmark (P-2021-541), including an approval to contact families outside the clinic (R-21041716). As a non-interventional observational study, it did not require additional ethical approval from the ethics committee.
HUMAN AND ANIMAL RIGHTS
All procedures performed in studies involving human participants were in accordance with the ethical standards of institutional and/or research committee and with the 1975 Declaration of Helsinki, as revised in 2013.
CONSENT FOR PUBLICATION
Participants aged 15 years and older provided informed consent before inclusion in the study, while parents or legal guardians provided consent for children under 15 years of age.
AVAILABILITY OF DATA AND MATERIAL
All data generated or analyzed during this study are included in this published article.
ACKNOWLEDGEMENTS
Declared none.
